Taubes makes a number of arguments in his response to my piece that merit dissection, but for the sake of being concise I will respond only to those that are the most important. Let’s focus on the primary bone of contention first, which is Taubes’s extraordinary claim about sugar. The claim in question is that sugar is the primary cause of obesity and common noncommunicable diseases such as cardiovascular disease and diabetes, and that other causes such as calorie intake, physical activity, and other aspects of diet quality are not important.
Taubes states that “If the sugar hypothesis is wrong, it is critically important that it be refuted definitively.” It is not the responsibility of the scientific community to refute every hypothesis that emerges from the popular press; if Taubes believes his extraordinary claim is correct, the onus is on him to provide extraordinary evidence in support of it. What is his evidence?
Taubes’s first piece of “evidence” is the assumption that obesity and most noncommunicable diseases are caused by one factor. This is based on a tool of logic called Occam’s Razor. Allow me to explain how Occam’s Razor works: All else being equal, the simpler of two hypotheses is most likely to be correct. The key phrase here is “all else being equal.” Occam’s Razor is a tie breaker at best, not a trump card that can be used to discard experimental evidence that challenges one’s hypothesis.
Having established that Occam’s Razor cannot be used to discard evidence, let’s consider what this evidence currently suggests about sugar:
- Sugar can increase body fatness, and this depends entirely on its ability to increase calorie intake.(1)
- A high intake of sugar does not impede weight loss when calorie intake is reduced.(2)
- Realistic doses of sugar have a modest negative impact on markers of diabetes risk, and this is potentiated by excess calorie intake.(3)
- Realistic doses of sugar have a modest negative impact on markers of cardiovascular disease risk.(4)
- Sugar is not very fattening in animal models of obesity, and its effects on body fatness and insulin resistance are generally less than those of added fat alone; the combination of sugar and fat is more harmful than either in isolation.(5–7)
- Various forms of sugar intake are often associated with weight gain in observational studies, among other factors, but the associations are inconsistent and generally not strong.(1)
- Refined sugar and/or sugar-sweetened beverage intake is associated with coronary heart disease risk, among many other factors such as low physical activity level, low fruit and vegetable intake, low nut intake, low dairy intake, and dietary fat quality.(8–11)
In aggregate, this evidence does not lend itself to the hypothesis that sugar is the primary driver of obesity or noncommunicable disease risk, although it does support the conventional hypothesis that refined sugar plays a role in obesity and noncommunicable disease, among other factors.
Taubes states that “because obesity and diabetes, particularly type 2, are intimately linked to insulin resistance, we should be looking ultimately and desperately for the cause of insulin resistance.” While Taubes continues his quest to uncover the causes of insulin resistance and type 2 diabetes, the scientific community has already identified them: Excess body fatness, physical inactivity, and genetics (12–19). This fact is supported by multiple long-term human trials demonstrating that even modest weight loss via calorie restriction and exercise reduces the incidence of type 2 diabetes by more than half (12–17). No such evidence exists for sugar.
Here is more evidence demonstrating that the factors Taubes believes to be unimportant are in fact central to insulin resistance and type 2 diabetes:
- Increasing calorie intake increases body weight, and this produces marked insulin resistance (20,21).
- Conversely, decreasing calorie intake decreases body weight and this substantially decreases insulin resistance (22).
- A long-term primate study suggests that calorie restriction markedly reduces the risk of diabetes and cardiovascular disease over the course of the lifespan (23). Human studies suggest that the same is likely true for us (24).
- Physical inactivity rapidly produces marked insulin resistance, and physical activity rapidly reverses it (25,26).
Rather than providing extraordinary evidence to support his extraordinary claims about sugar, Taubes offers weak evidence while omitting strong evidence to the contrary. So where, exactly, do his bold opinions on sugar come from? As it turns out, they come in large part from the very form of evidence he decries in his latest piece.
In response to my graph demonstrating that obesity has increased in the United States even as sugar intake has decreased, Taubes states “It’s this kind of simplistic argument and a lack of understanding of the very obvious limitations of observational and epidemiological data that I have argued in my books got us into this dire public health situation.” This statement is rather interesting since observational evidence is at the foundation of his own argument about sugar, and he cites observational evidence repeatedly in his books and in the very piece from which I drew that quote.[1]
A key argument of Taubes’s latest book, The Case Against Sugar, as well as his previous two books and his current response, is that observational evidence from cultures around the globe can tell us which factors are responsible for obesity and noncommunicable disease, and which factors aren’t. Here is how Taubes describes the argument in his current response: “I believe these historical populations, enumerated in my second book, Why We Get Fat, can be used to rule out suspected causes including ultraprocessed food, however that’s defined.”
This type of population-level observational evidence is called “ecological evidence.” Apparently Taubes considers it sufficiently robust to rest his own beliefs upon, yet when others cite similar evidence they are failing to appreciate its “very obvious limitations,” and they may even be precipitating a public health crisis.
If ecological evidence is admissible, as Taubes’s writing suggests it is, then perhaps we should see if there are any cultures that refute his assertion that obesity and chronic disease pop up anytime a high intake of sugar does. These are not difficult to find: The Hadza, a well-studied hunter-gatherer tribe in Tanzania, has a year-round average sugar intake from honey and fruit that approximates that of Americans (27). Yet they show no sign of obesity, diabetes, cardiovascular disease, or any of the other conditions Taubes attributes to sugar (28).
It is rather unclear to me, and to others who are familiar with the scientific literature on insulin resistance, obesity, chronic disease, and nutrition, how Taubes arrived at such an extreme position. The best I can gather from his most recent book and his writing here is that his belief originates primarily from Occam’s Razor and observational evidence. If Taubes has extraordinary evidence supporting his extraordinary claim about sugar, then perhaps this would be a good time to share it.
The Infamous Graph
In my initial response to Taubes, I presented evidence that sugar intake has been declining since 1999, while obesity and diabetes rates have continued to increase (this decrease in sugar intake is supported by multiple independent lines of evidence) (29–33). Taubes focused on the axes of the graph I presented, suggesting that the scale is misleading because it exaggerates the magnitude of the change in sugar intake. Allow me to restate what I explained in the graph’s legend: the purpose of the choice of scale was not to illustrate the magnitude of the changes in question, but rather to illustrate the correlation between obesity and sugar intake over time. Thus, I selected a scale that would allow a general audience to easily see the correlation of the two factors pre-1999, and the divergence post-1999. See below:
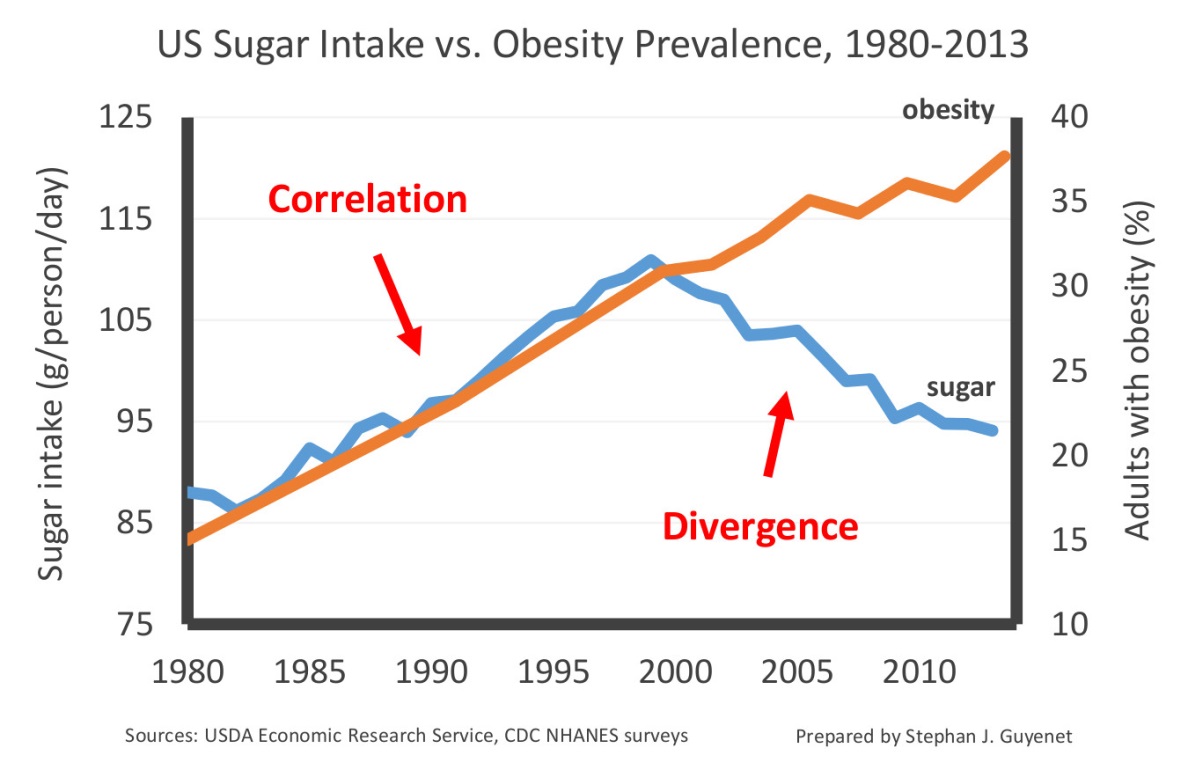
Taubes’s suggestion that I was attempting to exaggerate the decline in sugar intake is also difficult to understand because I stated its magnitude in the text: 18 percent. For reasons that are unclear, he shrinks this number to “a modest drop of 15 percent” in his response. Other sources indicate that sugar intake may actually have declined by up to 23 percent, and that sugar intake is either stable or decreasing in most countries globally even as obesity and diabetes rates continue to climb (34).
In any case, focusing on how I chose to construct my graph is a diversion from the real crux of the argument: The observation that sugar intake has been declining since 1999 and obesity and diabetes rates have not only failed to decrease, but continued to climb, is difficult to reconcile with the hypothesis that sugar is the primary cause of these conditions. Yet Taubes nimbly evades falsification by suggesting that the sugar we ate decades ago may be continuing to fatten us today. Does it matter that there is precisely no evidence to support this claim?[2] I happen to think so.
The Decline of Heart Attack Risk: Fact or Fiction?
Taubes does not attempt to defend his initial assertion that sugar is the primary cause of coronary heart disease. Instead, he argues that my piece attempted to “refute the claim that consuming sugar might cause heart disease,” and he disputes the evidence cited. Allow me to point out that I clearly stated in my piece that refined sugar likely contributes to coronary heart disease, and that the evidence I cited “doesn’t exonerate sugar.”
The issue at hand is not the uncontroversial proposition that refined sugar contributes to coronary heart disease. It is Taubes’s extraordinary claim that sugar is the primary cause of this disease, a claim for which he has provided no credible evidence.
Taubes does not feel sufficiently compelled by the evidence I presented demonstrating that coronary heart disease mortality has declined by more than 60 percent over the same period of time that sugar intake has increased. He notes correctly that mortality data can be confounded by advances in medicine. As I explained in my initial piece, advances in medicine are insufficient to account for this change, but even so, let’s consider a harder form of evidence: Autopsy data. This directly measures the presence of atherosclerosis in the coronary arteries, which is the definition of coronary heart disease.
At least two independent autopsy studies have been conducted over the time period in question, and both reported the same principal finding: Coronary artery disease decreased during the 1980s and 1990s as sugar intake was increasing (35,36). It is particularly telling that this decrease was also observed among younger people who were unlikely to be receiving medical therapy for heart disease.
This decline in coronary heart disease is not hard to explain. It is consistent with the decline of cigarette smoking and favorable changes in blood lipids, two well-established risk factors. (37) As Taubes notes, increases of obesity and diabetes may reverse this trend due to their impact on cardiovascular risk factors. Yet there is no evidence that sugar is the primary driver of this.
The Changing American Diet
In his response, Taubes cites observational data on U.S. food intake to support his contention that government dietary advice influenced American eating habits. Given his distaste for the “very obvious limitations” of observational evidence, it is unclear why he assumes that this association implies a causal effect of dietary advice on American eating habits.
It is true that the American diet has changed substantially over the course of the last 50 years, but these changes are not as easy to attribute to “dietary dogma” as Taubes assumes. In fact, the food and agriculture industries underwent substantial changes over the period in question that likely contributed to these shifts. For example, the inflation-adjusted cost of poultry plummeted, as did the cost of refined seed oils such as soybean oil. At the same time, the rise of industrially processed foods increased demand for cheap, stable, liquid fats such as soybean oils. Hydrogenated fats provided a cheaper and more stable alternative to lard and butter. In this context, it is not surprising that chicken and seed oils became more prevalent, at the expense of more costly and less convenient foods like butter, lard, and beef. It is certainly possible that cautions about saturated fat and cholesterol contributed to these changes, but this is not so clear since absolute saturated fat and cholesterol intake did not actually decline over the period in question.(29)
In any case, these details are a distraction from the main thrust of Taubes’s argument: dietary fat was “demonized” and sugar was “ignored,” and this explains why we eat so much sugar. As explained in my previous piece, dietary fat intake has not declined at all (not even “marginally” as Taubes states), and government dietary advice consistently recommended limiting sugar intake. These facts are difficult to reconcile with Taubes’s narrative.
Taubes implies that misguided advice from researchers and public health officials explains the fact that grain-based desserts like cake are the number one source of calories in the United States. As I explained in my last piece, we don’t eat cake and pizza because we’ve been snookered by researchers into thinking they will trim our waistlines. We eat them because we like them, despite the known consequences. Taubes continues to blame our junk food habit on the research and public health communities when common sense suggests otherwise.
Wrath of the Guidelines Redux
Taubes states that in his initial essay, he was not simply arguing that the USDA Dietary Guidelines caused us to eat more sugar or contributed to the epidemics of obesity and diabetes, but that the Guidelines were part of a broader “movement by the research community to demonize fat.” I accept the premise that his argument was not focused exclusively on the Guidelines, however the Guidelines clearly played an important part in it, as demonstrated by the first paragraph of his essay and a sentence from the second paragraph:
Forty years ago this month, January 1977, the federal government entered the business of giving dietary advice, first with a Congressional report released by George McGovern’s bipartisan Select Committee on Hunger and Human Needs, which in turn prompted the first Dietary Guidelines from the U.S. Department of Agriculture… While McGovern, his colleagues, and their committee staff may have all hoped the guidelines would lead to a nation eating, perhaps, as Michael Pollan would later memorably put it, (whole) food, “mostly plants and not too much,” that would not be the case.
Taubes asserts that the scientific and public health communities “demonized” fat and “ignored” sugar, a black and white narrative that does not fit the facts. As I demonstrated in my previous response, this bold assertion is not consistent with the equal attention paid to fat and sugar in the 1980 Guidelines—the document that was the primary interface between government dietary policy and the public. The Guidelines recommended that Americans “avoid too much” of both, a statement that does not demonize or ignore either one.
In any case, although history is not as black and white as Taubes suggests, I agree that the general cultural climate was to overemphasize dietary fat in the 1960s through the 1990s, at the expense of other important factors. The scientific and public health communities learned the hard way not to place undue emphasis on one nutritional factor, particularly in the absence of definitive evidence. I hope Taubes will not drive the U.S. public to repeat this mistake of history.
[1] For example, the observation that government diet recommendations coincided with changes in the U.S. diet. This association is assumed to be evidence of a cause-and-effect relationship between the two.
[2] Taubes cites evidence that certain health effects (e.g., lung cancer) can be separated from the exposure (e.g., cigarette smoke) by decades. But establishing that a delayed effect is biologically possible is not the same thing as establishing that it actually occurs for the particular exposure in question. Taubes cites evidence of intergenerational effects of obesity and metabolic disease, but these experiments are not relevant to the effects of sugar specifically.
Sources
1. Te Morenga L, Mallard S, Mann J. “Dietary sugars and body weight: systematic review and meta-analyses of randomised controlled trials and cohort studies.” BMJ. 2012 Jan 15;346:e7492.
2. Surwit RS, Feinglos MN, McCaskill CC, Clay SL, Babyak MA, Brownlow BS, et al. “Metabolic and behavioral effects of a high-sucrose diet during weight loss.” Am J Clin Nutr. 1997 Apr;65(4):908–15.
3. Horst KW ter, Schene MR, Holman R, Romijn JA, Serlie MJ. “Effect of fructose consumption on insulin sensitivity in nondiabetic subjects: a systematic review and meta-analysis of diet-intervention trials.” Am J Clin Nutr. 2016 Dec 1;104(6):1562–76.
4. Te Morenga LA, Howatson AJ, Jones RM, Mann J. “Dietary sugars and cardiometabolic risk: systematic review and meta-analyses of randomized controlled trials of the effects on blood pressure and lipids.” Am J Clin Nutr. 2014 Jul;100(1):65–79.
5. Surwit RS, Feinglos MN, Rodin J, Sutherland A, Petro AE, Opara EC, et al. “Differential effects of fat and sucrose on the development of obesity and diabetes in C57BL/6J and A/J mice.” Metabolism. 1995 May;44(5):645–51.
6. Sumiyoshi M, Sakanaka M, Kimura Y. “Chronic intake of high-fat and high-sucrose diets differentially affects glucose intolerance in mice.” J Nutr. 2006 Mar;136(3):582–7.
7. Lozano I, Van der Werf R, Bietiger W, Seyfritz E, Peronet C, Pinget M, et al. “High-fructose and high-fat diet-induced disorders in rats: impact on diabetes risk, hepatic and vascular complications.” Nutr Metab. 2016;13:15.
8. Fung TT, Malik V, Rexrode KM, Manson JE, Willett WC, Hu FB. “Sweetened beverage consumption and risk of coronary heart disease in women.” Am J Clin Nutr. 2009 Apr;89(4):1037–42.
9. de Koning L, Malik VS, Kellogg MD, Rimm EB, Willett WC, Hu FB. “Sweetened beverage consumption, incident coronary heart disease, and biomarkers of risk in men.” Circulation. 2012 Apr 10;125(14):1735–41, S1.
10. Yang Q, Zhang Z, Gregg EW, Flanders WD, Merritt R, Hu FB. “Added sugar intake and cardiovascular diseases mortality among US adults.” JAMA Intern Med. 2014 Apr;174(4):516–24.
11. Willett MDWC, Skerrett PJ. Eat, Drink, and Be Healthy: The Harvard Medical School Guide to Healthy Eating. 1 edition. New York: Free Press; 2005.
12. Knowler WC, Barrett-Connor E, Fowler SE, Hamman RF, Lachin JM, Walker EA, et al. “Reduction in the incidence of type 2 diabetes with lifestyle intervention or metformin.” N Engl J Med. 2002 Feb 7;346(6):393–403.
13. Hamman RF, Wing RR, Edelstein SL, Lachin JM, Bray GA, Delahanty L, et al. “Effect of weight loss with lifestyle intervention on risk of diabetes.” Diabetes Care. 2006 Sep;29(9):2102–7.
14. Tuomilehto J, Lindström J, Eriksson JG, Valle TT, Hämäläinen H, Ilanne-Parikka P, et al. “Prevention of type 2 diabetes mellitus by changes in lifestyle among subjects with impaired glucose tolerance.” N Engl J Med. 2001 May 3;344(18):1343–50.
15. Ramachandran A, Snehalatha C, Mary S, Mukesh B, Bhaskar AD, Vijay V, et al. “The Indian Diabetes Prevention Programme shows that lifestyle modification and metformin prevent type 2 diabetes in Asian Indian subjects with impaired glucose tolerance (IDPP-1).” Diabetologia. 2006 Feb;49(2):289–97.
16. Kosaka K, Noda M, Kuzuya T. “Prevention of type 2 diabetes by lifestyle intervention: a Japanese trial in IGT males.” Diabetes Res Clin Pract. 2005 Feb;67(2):152–62.
17. Pan XR, Li GW, Hu YH, Wang JX, Yang WY, An ZX, et al. “Effects of diet and exercise in preventing NIDDM in people with impaired glucose tolerance. The Da Qing IGT and Diabetes Study.” Diabetes Care. 1997 Apr;20(4):537–44.
18. Chan JM, Rimm EB, Colditz GA, Stampfer MJ, Willett WC. “Obesity, fat distribution, and weight gain as risk factors for clinical diabetes in men.” Diabetes Care. 1994 Sep;17(9):961–9.
19. Poulsen P, Kyvik KO, Vaag A, Beck-Nielsen H. “Heritability of type II (non-insulin-dependent) diabetes mellitus and abnormal glucose tolerance—a population-based twin study.” Diabetologia. 1999 Feb;42(2):139–45.
20. Erdmann J, Kallabis B, Oppel U, Sypchenko O, Wagenpfeil S, Schusdziarra V. “Development of hyperinsulinemia and insulin resistance during the early stage of weight gain.” Am J Physiol Endocrinol Metab. 2008 Mar;294(3):E568-575.
21. Sims E a. H, Horton ES. “Endocrine and Metabolic Adaptation to Obesity and Starvation.” Am J Clin Nutr. 1968 Dec 1;21(12):1455–70.
22. Larson-Meyer DE, Heilbronn LK, Redman LM, Newcomer BR, Frisard MI, Anton S, et al. “Effect of calorie restriction with or without exercise on insulin sensitivity, beta-cell function, fat cell size, and ectopic lipid in overweight subjects.” Diabetes Care. 2006 Jun;29(6):1337–44.
23. Colman RJ, Anderson RM, Johnson SC, Kastman EK, Kosmatka KJ, Beasley TM, et al. “Caloric restriction delays disease onset and mortality in rhesus monkeys.” Science. 2009 Jul 10;325(5937):201–4.
24. Fontana L, Meyer TE, Klein S, Holloszy JO. “Long-term calorie restriction is highly effective in reducing the risk for atherosclerosis in humans.” Proc Natl Acad Sci U S A. 2004 Apr 27;101(17):6659–63.
25. Hamburg NM, McMackin CJ, Huang AL, Shenouda SM, Widlansky ME, Schulz E, et al. “Physical inactivity rapidly induces insulin resistance and microvascular dysfunction in healthy volunteers.” Arterioscler Thromb Vasc Biol. 2007 Dec;27(12):2650–6.
26. Zierath JR. “Invited review: Exercise training-induced changes in insulin signaling in skeletal muscle.” J Appl Physiol Bethesda Md 1985. 2002 Aug;93(2):773–81.
27. Marlowe FW, Berbesque JC, Wood B, Crittenden A, Porter C, Mabulla A. “Honey, Hadza, hunter-gatherers, and human evolution.” J Hum Evol. 2014 Jun;71:119–28.
28. Raichlen DA, Pontzer H, Harris JA, Mabulla AZP, Marlowe FW, Josh Snodgrass J, et al. “Physical activity patterns and biomarkers of cardiovascular disease risk in hunter-gatherers.” Am J Hum Biol Off J Hum Biol Counc. 2016 Oct 9.
29. USDA Economic Research Service - Food Availability (Per Capita) Data System [Internet]. 2016 [cited 2016 Dec 22]. Available from: https://www.ers.usda.gov/data-products/food-availability-per-capita-data-system/
30. Fryar CD, Carroll MD, Ogden CL. “Prevalence of Overweight, Obesity, and Extreme Obesity Among Adults Aged 20 and Over: United States, 1960–1962 Through 2013–2014.” Natl Cent Health Stat Health E-Stats. 2016 Jul.
31. Menke A, Casagrande S, Geiss L, Cowie CC. “Prevalence of and Trends in Diabetes Among Adults in the United States, 1988-2012.” JAMA. 2015 Sep 8;314(10):1021–9.
32. Kit BK, Fakhouri THI, Park S, Nielsen SJ, Ogden CL. “Trends in sugar-sweetened beverage consumption among youth and adults in the United States: 1999-2010.” Am J Clin Nutr. 2013 Jul;98(1):180–8.
33. Makarem N, Scott M, Quatromoni P, Jacques P, Parekh N. “Trends in dietary carbohydrate consumption from 1991 to 2008 in the Framingham Heart Study Offspring Cohort.” Br J Nutr. 2014 Jun;111(11):2010–23.
34. Wittekind A, Walton J. “Worldwide trends in dietary sugars intake.” Nutr Res Rev. 2014 Dec;27(2):330–45.
35. Roger VL, Weston SA, Killian JM, Pfeifer EA, Belau PG, Kottke TE, et al. “Time trends in the prevalence of atherosclerosis: a population-based autopsy study.” Am J Med. 2001 Mar;110(4):267–73.
36. Nemetz PN, Roger VL, Ransom JE, Bailey KR, Edwards WD, Leibson CL. “Recent trends in the prevalence of coronary disease: a population-based autopsy study of nonnatural deaths.” Arch Intern Med. 2008 Feb 11;168(3):264–70.
37. Ford ES, Ajani UA, Croft JB, Critchley JA, Labarthe DR, Kottke TE, et al. “Explaining the decrease in U.S. deaths from coronary disease, 1980-2000.” N Engl J Med. 2007 Jun 7;356(23):2388–98.

